2023-03-28 マサチューセッツ工科大学(MIT)
研究者らは、セメント製造に伴う二酸化炭素の総量の最大15%を、コンクリートの混合と打設の初期段階で無機化できることを実証しました。
その結果、新しい複合相が形成され、コンクリートがより早く固まるので、建設業界の生産性が向上します。この発見は、カーボンニュートラルな建設資材の開発に明るい展望をもたらすものです。
<関連情報>
- https://news.mit.edu/2023/new-additives-concrete-effective-carbon-sink-0328
- https://academic.oup.com/pnasnexus/article/2/3/pgad052/7089570?login=false
CO2をC-S-Hに固める:コンクリートカーボンニュートラルへの一歩 Cementing CO2 into C-S-H: A step toward concrete carbon neutrality
Damian Stefaniuk, Marcin Hajduczek, James C Weaver, Franz J Ulm, Admir Masic
PNAS Nexus Published:28 March 2023
DOI:https://doi.org/10.1093/pnasnexus/pgad052
Abstract
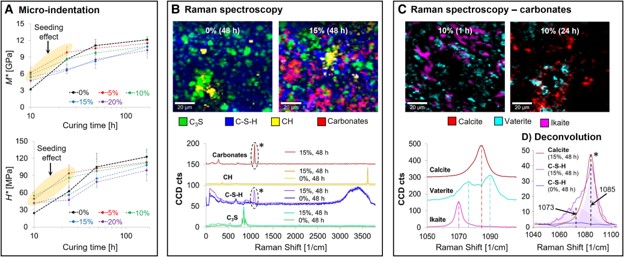
Addressing the existing gap between currently available mitigation strategies for greenhouse gas emissions associated with ordinary Portland cement production and the 2050 carbon neutrality goal represents a significant challenge. In order to bridge this gap, one potential option is the direct gaseous sequestration and storage of anthropogenic CO2 in concrete through forced carbonate mineralization in both the cementing minerals and their aggregates. To better clarify the potential strategic benefits of these processes, here, we apply an integrated correlative time- and space-resolved Raman microscopy and indentation approach to investigate the underlying mechanisms and chemomechanics of cement carbonation over time scales ranging from the first few hours to several days using bicarbonate-substituted alite as a model system. In these reactions, the carbonation of transient disordered calcium hydroxide particles at the hydration site leads to the formation of a series of calcium carbonate polymorphs including disordered calcium carbonate, ikaite, vaterite, and calcite, which serve as nucleation sites for the formation of a calcium carbonate/calcium-silicate-hydrate (C-S-H) composite, and the subsequent acceleration of the curing process. The results from these studies reveal that in contrast to late-stage cement carbonation processes, these early stage (precure) out-of-equilibrium carbonation reactions do not compromise the material’s structural integrity, while allowing significant quantities of CO2 (up to 15 w%) to be incorporated into the cementing matrix. The out-of-equilibrium carbonation of hydrating clinker thus provides an avenue for reducing the environmental footprint of cementitious materials via the uptake and long-term storage of anthropogenic CO2.